|
‘This work confronts a particularly tricky problem,’ says Susan Perkin from the University of Oxford, UK. ‘At the nanoscale the atomic interaction becomes important – do the water molecules stick or slip on the surface of the container?’ Hydrophobic materials are slippier and so the water flows more easily, appearing to be less viscous. ‘In a hydrophobic material, the energy to move a single molecule on the surface is lower than in a hydrophilic material,’ says Riedo. ‘Depending on which material we used, we saw pretty dramatic changes in viscosity,’ Riedo explains – on hydrophobic materials, the process required less force, meaning the water was less viscous. To confirm this, Riedo measured the viscosity of water on surfaces of differing hydrophobicity by dragging an atomic force microscope tip through a layer of water just five molecules thick. The team predicted that hydrophobic materials that have lower wettability, on which water spreads less easily, would lead to less viscous water. We then looked into the chemistry behind the confining surfaces.’ ‘We knew the viscosity of water was different at the nanoscale, it was more like ketchup. Riedo and her team were investigating whether nanoscale volumes of water, such as those found in our cell membranes, diffuse in the same way as bulk water. Now, Elisa Riedo and others at the Georgia Institute of Technology have found a relationship between this apparent change in viscosity and the water–surface interaction. It is well known that liquids confined between hydrophilic surfaces only a few nanometres apart exhibit a large increase in viscosity compared to the bulk, but researchers have struggled to understand why. The effect is only observed at the nanoscale and could lead to a deeper understanding of cell membrane transport, nanofluidics and advances in high resolution two- and three-dimensional printing. Shear Viscosity Range: 0.Scientists in the US have discovered that water’s viscosity is affected by the material its container is made from.microVISC™ simplicity and fast operation capabilities make it the ideal candidate for QC viscosity measurements. Additionally, these measurements demonstrate the need to characterize each specific formulation viscosity as oppossed to interpolating viscosity values based on pure component's properties. microVISC™ results show excellent agreement with reference values obtained using alternative techniques. In this application note we present a study of the effect of concentration on viscosity for volume fractions ranging between both pure components. 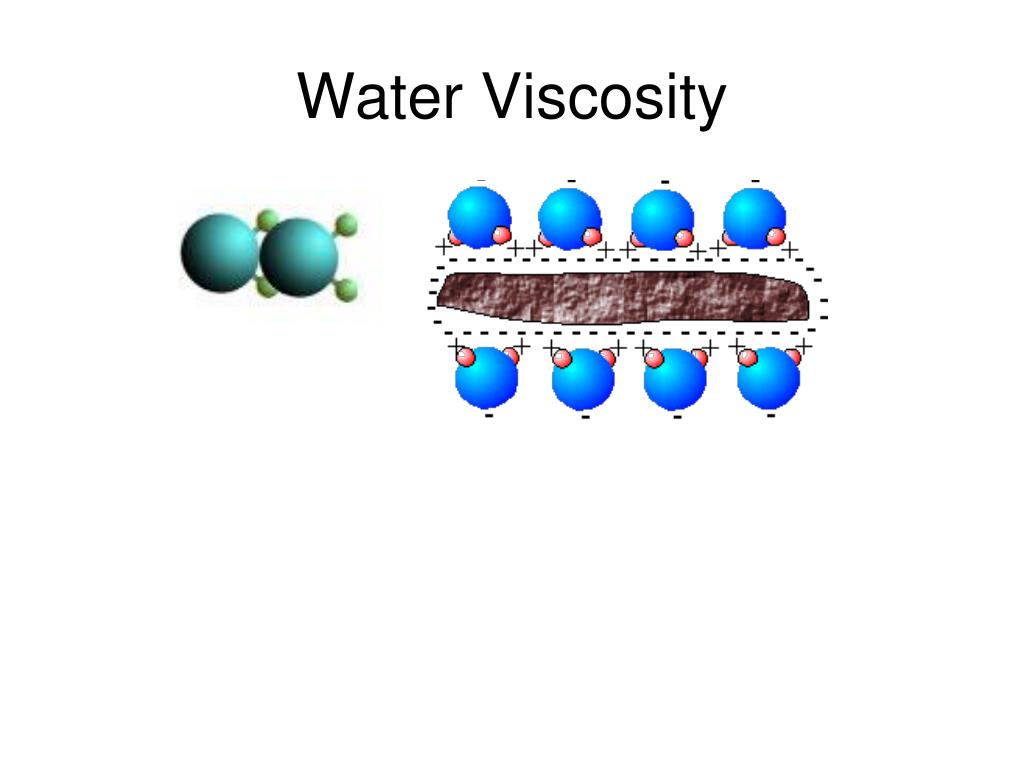
a small change in concentration can result in a dramatic and non-linear change in properties such as viscosity) making accurate measurements essential. However, these compounds behave like non-ideal mixtures (i.e. Modeling and quality control of industrial manufacturing processes require proper characterization of viscosity. Water-Alcohol mixtures are widely used in chemical engineering applications such as solvents for gums, resins, lacquers, varnishes and dyes. Viscosity of Two Component Mixtures Viscosity of Water-Ethanol and Water-Isopropyl Alcohol Mixtures
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
